Enrolling Now
Enrolling Now
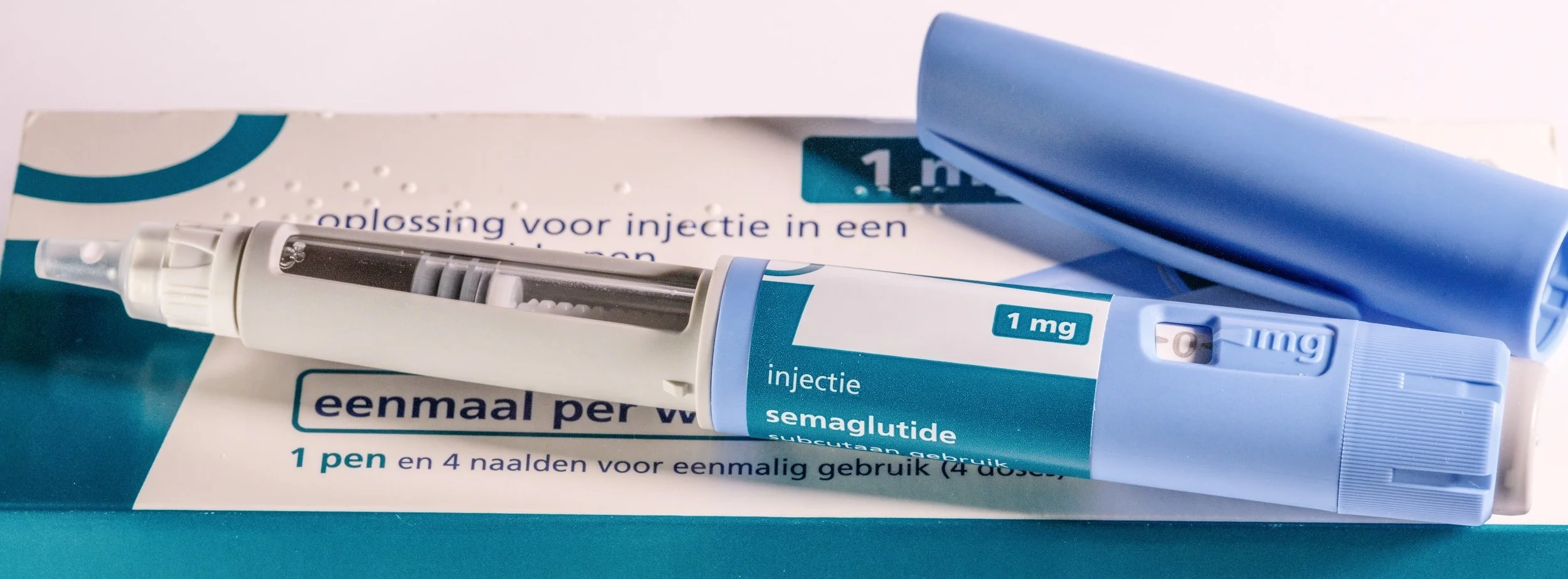
Subcutaneous Medication for HbA1c
-
A study to evaluate a subcutaneous medication in participants with T2D on a stable dose of Tirzepatide or Semaglutide who failed to achieve HbA1c goal.
-
No Type 1 Diabetes
Not on insulin
18-75 years of age
T2D with HbA1c >7.5% to <10.5%
BMI of >27 kg/m2, with stable body weight for at least 3 months.
Have been on stable dose of either injectable semaglutide or tirzepatide for >3 months
Further qualifications may apply.
-
Gain access to cutting-edge medications and treatments before they are widely available.
Our expert team is under the guidance of renowned endocrinologist, Dr. Anastasios Manessis.
We are highly focused on patient well-being and the highest ethical standards in clinical research.
Think you may qualify? Fill out this form and a member of our medical research team will be in touch shortly with more information.